Blog
The Criticality of Environmental Sampling

Although the United States is known for having one of the safest food and beverage supplies globally, illnesses with a foodborne base persist in the country. These foodborne illnesses affect millions and cause the deaths of thousands of people. Severe outbreaks weaken consumer confidence in affected products, lessen market demand, and instigate extensive recalls. Effective and thorough environmental sampling is a critical weapon in mitigating the effects of food contamination as it encompasses the raw ingredients, the process itself, and the processing environment.
Indicator Organisms
Indicator organisms are non-pathogenic microorganisms, such as aerobic plate count and total coliform, that may exist naturally in food and the environment. In general, indicator organisms are more abundant than pathogens, and detection of them is easier and quicker. Plus, their log reduction is regarded as a log reduction in pathogenic risk as well. They are employed to evaluate the inclusive environmental or sanitation state that could warn of the possible presence of pathogens. Food manufacturers should test all production zones for indicator organisms. Zone 1, the food contact zone, is investigated for indicator organisms because their detection allows for remedial steps and improvement without necessarily causing product holds or recalls.
The advantages of indicators organisms include:
- Cheaper and easier to detect and save time compared to pathogens.
- More plentiful than pathogens ensuring that control and remedial steps are measurable. The low amount of pathogens limits the significance of pathogen testing.
- Exist in high quantities and can be easily numbered.
- A legitimate representative of pathogens because they use the same water, temperature, nutrients, and pH. They are often closely related, e.g., gram-negative of fecal origin.
Microbial Trends
Concerning pathogens contaminating food, the stakes have only gotten higher. Due to previous incidents, the production of foods, such as dried dairy products, roasted nuts, soft cheeses, and peanut butter, needs to have an environmental sampling and monitoring program. Of increasing concern are pathogens including Salmonella, Campylobacter, hemorrhagic Escherichia coli, Staphylococcus aureus, Clostridium perfringens, Giardia duodenalis, Vibrio cholera, Rotavirus, and Bacillus cereus.

Traditional methods for environmental evaluation include vector swabbing procedures, ATP/bioluminescence for food residues and microbes, and ELISA kits for the detection of allergens. After a century of testing for coliform in the U.S., the current trend is to test for a broader class of Enterobacteriaceae (EB), which encompasses coliform, but also other fecal pathogens such as Salmonella and Shigella.
What is Environmental Monitoring?
During inspections, investigators collect samples from a building environment of a food manufacturing plant to find out if there are any detrimental bacteria present. Inspectors take samples during a production run as well as post-cleaning and sanitation. This sampling can involve checking food debris, surfaces, water, and air, such as dust control. With environmental sampling, investigators can use swabs or sterile sponges to collect samples that involve food contact and non-food contact surfaces.

This inspection often involves testing for indicators as well as for pathogens, namely Salmonella, E. coli, and Listeria. Generally, it is a good idea to screen, correct, and control indicators before sampling for pathogens. Regarding a strategy for environmental monitoring, if indicators are not in control showing a higher probability of pathogen detection, why increase pathogen swabbing and risk a potential product recall?
Who Should be Monitoring?
Food processors and manufacturers, who need to develop data supporting their sanitation hygiene practices, their process run times, and their shelf life determinations, need to be monitoring. Environmental sampling and monitoring are considered integral pieces of a pathogen control program in a food facility. For Pathogen Environmental Monitoring (PEM), choose a team that includes those who can cover sanitation, maintenance, production, quality, and record-keeping plus consultants.
When and How Frequently to Sample?
Regarding the “when” of this question, environmental sampling can be done for cause that includes:
- When there is a sign of microbial contaminants being present, which might involve prior detection of environmental pathogens.
- Observations from inspections that necessitate taking samples for analysis.
- When performing extended product runs and an increase in indicators predicts it is a good time to end the run and clean.
- A follow-up to the discovery of a pathogen in a product sample.
Regarding frequency, this is an example of a customer determined testing schedule by production zone:
- For Zone 1: Weekly, post-cleaning pre-sanitizer application
- For Zone 2: Weekly
- For Zone 3: Weekly
- For Zone 4: Monthly
Post-Sanitizing Monitoring: For testing of sanitation effect on indicator organisms, take samples after cleaning and sanitization but before production startup.
Where to Test?
Zoning according to proximity to food categorizes where to test.
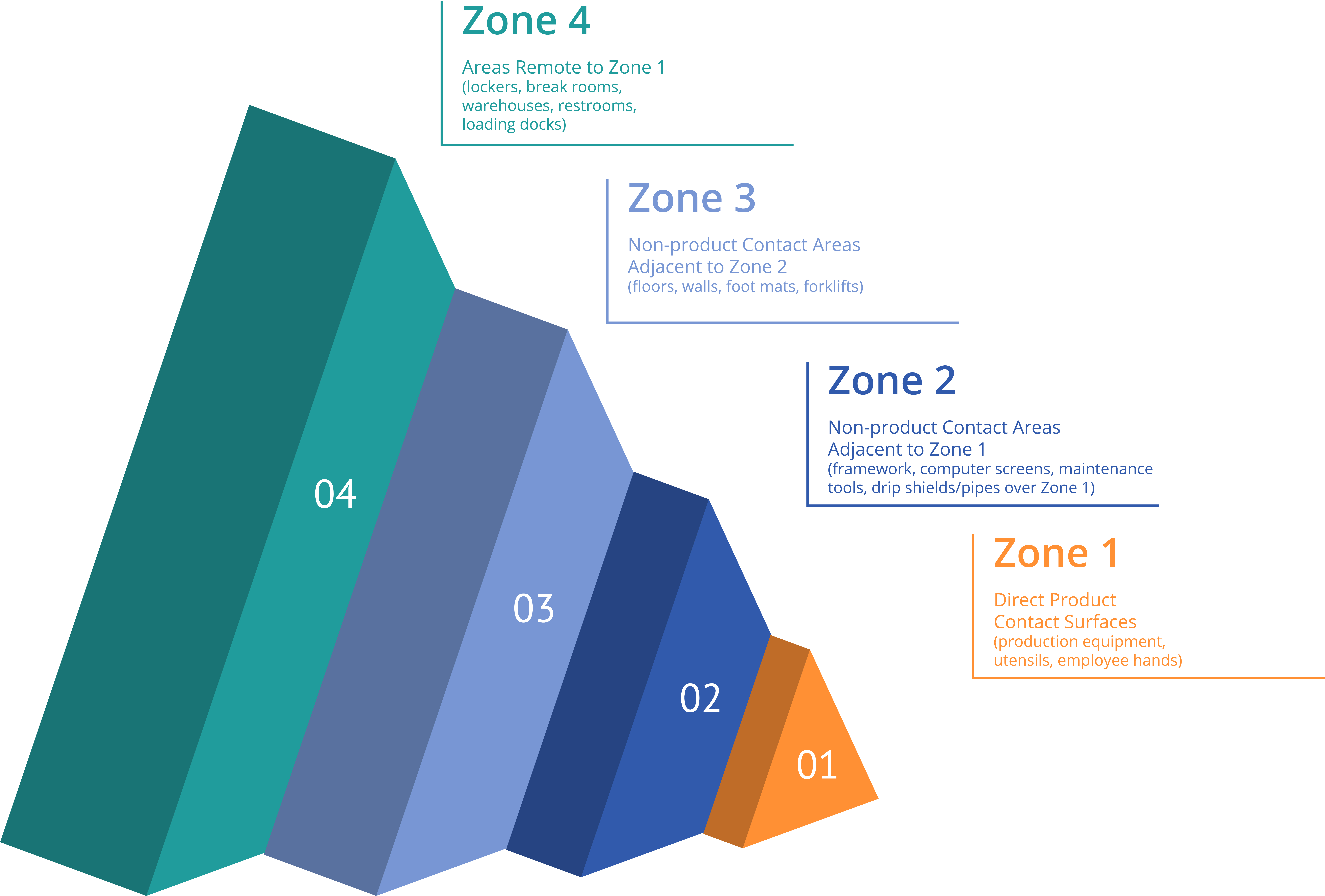
Zone 1: Direct product contact surfaces. Examples are conveyors/buckets, utensils, employee hands, slicers/pitters, and hoppers/bins/bin liners/fillers.
Zone 2: Non-product contact sites adjacent to Zone 1. Examples are equipment framework, drip shields/housing/control panels/buttons, pipes over Zone 1, computer screens, and maintenance tools.
Zone 3: Non-produce contact sites adjacent to Zone 2. Examples are floors/walls/ceilings, hoses/air handling units, drains, foot mats/baths, forklifts, brooms/mops, and pallets.
Zone 4: Areas remote to Zone 1. Examples are locker/break rooms/offices, warehouses/freezers/cold storage, restrooms, loading docks, and maintenance shop.
How to Test?
Sampling tools can include swabs (sponge and Q-tip style), sterile scoops, spatulas, and sample cups. The area sampled can vary between 40-200 in2 for indicators and 40-400 in2 for pathogens. If sending samples to a 3rd party lab for testing, transport the samples at less than 45 0 F and test within 48 hours.
Tests for indicator organisms can include aerobic plate count, coliforms, Enterobacteriaceae, and Yeast and Mold. Pathogen tests include Salmonella, Listeria, and E. coli, depending on the product risk.

Use the quantitative data from Zone 1 to evaluate sanitation programs.
Recommended Microbiological Indicator Limits for Equipment Cleaning Before and After Application of Sanitizer:
You are trying to load a table of an unknown type. Probably you did not activate the addon which is required to use this table type.
Environmental Swabbing Procedures:
Sponge Sampling
Grasping stick, press sponge to survey 100 cm2 surface. Return sampling device to the bag. Detach and dispose of the sponge handle. Secure sample bag. The sample is ready for microbial analysis.
To plate, squeeze the sponge to collect the liquid in the opposite corner of the bag. Pipet sample. Incubate at appropriate times and temperatures.
Swab Sampling
Scrub swab across a surface. Return swab to the tube. The sample is ready for microbial analysis.
To plate, shake the tube to extract the sample. Remove the swab from the tube, wringing out any extra buffer. Pipet sample from the tube. Incubate at the appropriate times and temperatures.
For air-flow and water/liquid filtration methods, consult the CDC document entitled “Infection Control.”
Monitor Trends
Historical data is required to establish a baseline. As an example, if a site tests <40 cfu for a year with three or four spike readings, then 40 cfu would be the baseline.
Why It Is Important
An environmental monitoring program is critical for these reasons:

- Gauge the overall effectiveness of operational methods, sanitary design, and personnel practices.
- Offers information including source and concentration concerning indicator organisms, spoilage organisms, and pathogens so that corrective actions help prevent microbial outbreaks.
- It authenticates the sanitation program and assists in determining the frequency needed for cleaning and sanitation.
- Functions as an early warning system for microbiological dangers in the production and post-production environment.
- Assists in locating harboring niches and hot spots in a plant, which could be sources of contamination.
- It is an important element of documenting the sanitary state of the facility. It can be useful in validating production times and determining when to end them and clean the lines.
- Determines if plant maintenance, such as the changing of filters and gaskets, is required.
- It can result in additional testing that might end up putting a product on hold.
- Functions as an essential training tool for workers.
Post-Sanitation
A pre-operational examination of the food processing facilities and equipment can assist in determining if cleaning and sanitation were effective. Standard Operating Procedures (SOP) contain written detailed instructions for routine operations at a facility. SOPs can cover items such as hand washing and cleaning and sanitizing food contact surfaces.
ATP/Bioluminescence can detect food residues and microbes. The absence of ATP on a surface is an indication of cleanliness. ATP testing is utilized during pre-operation inspections to verify that equipment is free from residual food and microbial contamination.

ELISA kits detect allergens on a surface. Residual allergen proteins on a surface can contaminate a product and prompt a food recall.
The Mission
Although so much attention is focused presently on the coronavirus pandemic, we should never lose sight of the fact that protecting the safety and integrity of our food and beverages requires constant vigilance and effort. The criticality of environmental sampling cannot be overstated. Effective environmental monitoring is an early warning system that gauges the performance of your food safety programs. It enables an organization to have important legal protection and demonstrates to customers and regulators that your organization is very serious about food safety.
How Charm Can Help
You can employ a preventative strategy to protect your brands from recalls by using environmental monitoring sampling, in conjunction with microbial indicator testing.
About Charm Sciences
Established in 1978 in Greater Boston, Charm Sciences helps protect consumers, manufacturers, and global brands from a variety of issues through the development of food safety, water quality, and environmental diagnostics tests and equipment. Selling directly and through its network of distributors, Charm’s products serve the dairy, feed and grain, food and beverage, water, healthcare, environmental, and industrial markets in more than 100 countries around the globe.
